行业质量体系在药品CGMP法规方面的指导原则(中文)
VIP免费
CGMP
CDER
CBER
CVM
ORA
2006 9
CGMP
CGMP
HFD-240
5600
20857
301-827-4573
http://www.fda.gov/cder/guidance/index.htm
HFM-40
1401
20852-1448 http://www.fda.gov/cber/guidelines.
htm.800-835-4709 301-827-180
HFV-12
7519 2085
301-827-380
http://www.fda.gov/cvm/guidance/published.html
CDER
CBER
CVMORA
2006 9 CGMP
Contains Nonbinding Recommendations
. ....................................................................................................................................... 1
II. ............................................................................................................................ 1
A. .....................................................................................................................................................................1
B. .........................................................................................................................................................2
C. .........................................................................................................................................................3
D. .............................................................................................................................................4
III. CGMPS ........................................................................................... 4
A. ......................................................................................................................................................................4
B. .............................................................................................................................4
C. .....................................................................................................................................................5
D. CAPA...............................................................................................................................5
E. .............................................................................................................................................................5
F. .............................................................................................................................................................5
G. ..................................................................................................................................................6
IV. ....................................................................................................................... 8
A. .............................................................................................................................................................8
1. .........................................................................................................................................................8
2. ............................................................................................................................................................9
3. .......................................................................................................................9
4. .....................................................................................................................................10
5. ...........................................................................................................................................................10
B. ...................................................................................................................................................................12
1. ...........................................................................................................................................................12
2. ...........................................................................................................................................................13
3. ........................................................................................................................................................13
4. ..................................................................................................................................................14
C. ..................................................................................................................................................................15
1. ..........................................................................................................................15
2. ..........................................................................................................................................................16
3. .............................................................................................................................................17
4. ...................................................................................................................................................19
D. ............................................................................................................................................................21
1. ......................................................................................................................................21
2. ....................................................................................................................................................21
3. ..................................................................................................................................................22
4. ............................................................................................................................................................22
摘要:
展开>>
收起<<
oTCGMPlvOSelLNcSWVSkuNQlOgRToTvwctoTNTxzvNCDERuriR6TO0xzvNCBERQoNCVMvNRRQlORA2006t9goTuNtCGMPoTCGMPlvOSelLNcSWYvRogSNNNnSSWNlRQloTOoYHFD240oTNTxzvNToTvwct95600SlQpWQK20857Su53018274573httpwwwfdagovcderguidanceindexhtmb0WNRQluriR6TO0xzvNHFM40R6UFSORToTvwct1401WQKYSWQKlQp208521448httpwwwfdagovcberguid...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
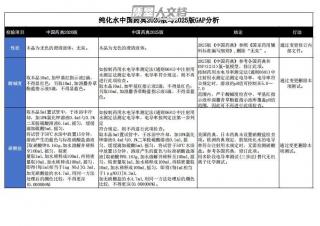
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 384
2025-04-18 384 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 611
2025-09-27 611 -
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 563
2025-09-29 563 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 999+
2025-09-29 999+ -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 521
2025-11-06 521 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 176
2025-11-06 176 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 186
2025-11-19 186 -
注射用药品的目视检查-第五版-2025.10_中文VIP免费

 2025-11-25 82
2025-11-25 82 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 439
2025-11-26 439 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 85
2026-03-19 85
作者:冒牌货
分类:专业资料
价格:150质量币
属性:32 页
大小:6.42MB
格式:PDF
时间:2025-12-04


