Q4B Annex 6 Uniformity of Dosage Units General ChapterQ4B附录6 统一剂量单位 常规篇
VIP免费
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL
REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE
ICH HARMONISED TRIPARTITE GUIDELINE
EVALUATION AND RECOMMENDATION OF PHARMACOPOEIAL
TEXTS FOR USE IN THE ICH REGIONS
ON
UNIFORMITY OF DOSAGE UNITS GENERAL CHAPTER
Q4B ANNEX 6
Current
Step 4
version
dated 13 November 2013
This Guideline has been developed by the appropriate ICH Expert Working Group
and has been subject to consultation by the regulatory parties, in accordance with the
ICH Process. At Step 4 of the Process the final draft is recommended for adoption to
the regulatory bodies of the European Union, Japan and USA.
Q4B Annex 6
Document History
Code
History
Date
Q4B Annex 6
Approval by the Steering Committee under
Step 2
and release
for public consultation.
13 November
2008
Q4B Annex
6(R1)
Integration into
Step 2
of the Health Canada
Interchangeability Statement under Section 4.5 after
approval by the Steering Committee.
27 September
2010
Current
Step 4
version
Q4B Annex 6
Approval by the Steering Committee under
Step 4
and
recommendation for adoption to the three ICH regulatory
bodies. The R was removed from the name of the document as
this is the first version of the Annex 6
Step 4.
13 November
2013
i
EVALUATION AND RECOMMENDATION OF PHARMACOPOEIAL
TEXTS FOR USE IN THE ICH REGIONS
ON
UNIFORMITY OF DOSAGE UNITS GENERAL CHAPTER
Q4B ANNEX 6
ICH Harmonised Tripartite Guideline
Having reached
Step 4
of the ICH Process at the ICH Steering Committee meeting on 13
November 2013, this guideline is recommended for adoption
to the three regulatory parties to ICH.
TABLE OF CONTENTS
1. INTRODUCTION...................................................................................................... 1
2. Q4B OUTCOME ........................................................................................................ 1
2.1 Analytical Procedures .................................................................................................... 1
2.2 Acceptance Criteria ....................................................................................................... 1
3. TIMING OF ANNEX IMPLEMENTATION .............................................................. 1
4. CONSIDERATIONS FOR IMPLEMENTATION ...................................................... 1
4.1 General Consideration ................................................................................................... 1
4.2 FDA Consideration ........................................................................................................ 1
4.3 EU Consideration .......................................................................................................... 2
4.4 MHLW Consideration .................................................................................................... 2
4.5 Health Canada Consideration ....................................................................................... 2
5. REFERENCES USED FOR THE Q4B EVALUATION ............................................. 2
标签: #附录
摘要:
展开>>
收起<<
INTERNATIONALCONFERENCEONHARMONISATIONOFTECHNICALREQUIREMENTSFORREGISTRATIONOFPHARMACEUTICALSFORHUMANUSEICHHARMONISEDTRIPARTITEGUIDELINEEVALUATIONANDRECOMMENDATIONOFPHARMACOPOEIALTEXTSFORUSEINTHEICHREGIONSONUNIFORMITYOFDOSAGEUNITSGENERALCHAPTERQ4BANNEX6CurrentStep4versiondated13November2013ThisGuide...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
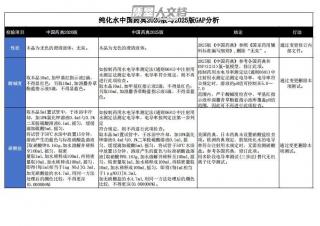
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 383
2025-04-18 383 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 608
2025-09-27 608 -
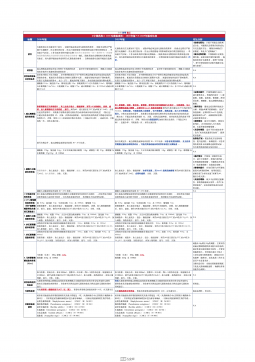
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 557
2025-09-29 557 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 999+
2025-09-29 999+ -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 520
2025-11-06 520 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 176
2025-11-06 176 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 186
2025-11-19 186 -
注射用药品的目视检查-第五版-2025.10_中文VIP免费

 2025-11-25 82
2025-11-25 82 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 438
2025-11-26 438 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 85
2026-03-19 85
作者:冒牌货
分类:专业资料
价格:50质量币
属性:6 页
大小:62.85KB
格式:PDF
时间:2025-12-24


