MDCG 2020-6 (欧盟)2017∕745号法规此前根据93∕42∕EEC指令或90∕385∕EEC指令获得CE标志的医疗器械所需临床证据
VIP免费
MDCG 2020-6
Medical Device Coordination Group Document
Medical Device
MDCG 2020-6
2017/745 93/42/EEC 90/
385/EEC CE
2020 4
2017/745 103 MDCG
MDCG
2017/745 93/42/EEC 90/385/EEC
CE
Page 2 of 22
1. ................................................................................................................................................4
1.1. MDR 2 ........................................................ 4
1.2. MDR 2 ...................................................................................... 4
2. .......................................................................................................................................6
3. ................................................................................................................................................6
4. ...................................................................................................................................7
5. .........................................................................................................................................8
6. ............................................................................... 10
6.1. XIV A 1a ...................................................... 10
6.2. XIV A 1b .............................................................. 11
6.2.1. ................................................................................................ 12
6.2.2. .................................................................................................. 12
6.3. XIV A 1c ......................................................................... 12
6.4. XIV A 1d .................................................................. 13
6.5. XIV A 1e .......................................................................... 14
I - MDR MEDDEV 2.7/1 rev. 4
............................................................................................................................. 18
....................................................................................................... 19
-
MDRGSPR ....................................... 20
Page 3 of 22
摘要:
展开>>
收起<<
MDCG20206MedicalDeviceCoordinationGroupDocumentMedicalDeviceMDCG20206kv2017745SlkdRMh9cn9342EECcNb90385EECcNCEhvSuVhhbN4cnR6UFTQlTJggcSW2020t4ggeNu1kv2017745Sl103gazvSuVhhSOMDCGS0MDCGu1bgbTXVvNhbu1kvYTXOvNTNhbNN0geNvkvYTXOeNNNSfkvYTXOvezW0geNNhvNOUpWGNQwglgRSgkvlbYkvlOQQwggRv0kv2017745SlkdRMh9cn9342...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
环境体系管理评审文件VIP免费

 2024-04-19 96
2024-04-19 96 -
管理评审控制程序(体系文件)VIP免费

 2024-04-19 106
2024-04-19 106 -
QP-05 管理评审控制程序VIP免费

 2024-04-19 150
2024-04-19 150 -
ISO17025:2017一整套程序文件(实验室认可服务)VIP免费

 2024-06-05 120
2024-06-05 120 -
医疗器械工作程序文件VIP免费
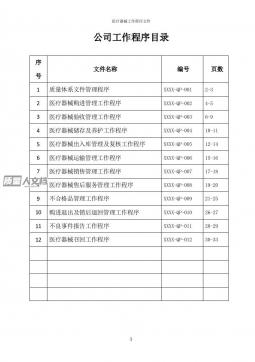
 2024-12-10 168
2024-12-10 168 -
确认与验证管理规程VIP免费

 2025-04-21 58
2025-04-21 58 -
有害物质管理程序(含表格)VIP免费

 2025-05-04 76
2025-05-04 76 -
医疗器械上市后临床跟踪控制程序VIP免费

 2025-10-27 51
2025-10-27 51 -
供应商质量管理程序VIP免费

 2025-11-07 105
2025-11-07 105 -
DC2-09001 环境因素识别、评价与控制程序VIP免费
 2026-03-21 18
2026-03-21 18
作者:一如既往
分类:法规规范
价格:120质量币
属性:22 页
大小:1.95MB
格式:PDF
时间:2026-03-17


