MDCG2020-10∕1修订版1 《欧盟法规(EU)2017745》下医疗器械临床研究中的安全性报告
MDCG 2020-10/1 1
EU2017/745
2022 10
2017/745 103 MDCGMDCG
Page 1 of 19
5.1
9.4
5.2.2
10.2.1
4
5
10.2.19 - 20
10.2.15 17
MDCG 2020-10/1 1
Page 2 of 19
May 2020
MDCG 2020-10 Rev 1
Safety reporting in clinical investigations
of medical devices under the Regulation (EU) 2017/745
1 ............................................................................................................................................................................................................................................................................................................ 3
1.1 EUDAMED ...................................................................................................................................................................................................................................................................3
2 ............................................................................................................................................................................................................................................................................................................ 4
2.1 ............................................................................................................................................................................................................................................................................................................4
2.2 ..................................................................................................................................................................................................................................................................4
3 ............................................................................................................................................................................................................................................................................................................ 5
3.1 ........................................................................................................................................................................................................................................................................................5
3.2 AE ............................................................................................................................................................................................................................................................................ 5
3.3 SAE .................................................................................................................................................................................................................................................................. 5
3.4 ............................................................................................................................................................................................................................................................................................5
4 ..................................................................................................................................................................................................................................................................................................... 6
4.1 EUDAMED .............................................................................................................................................................................................................................................................................6
4.2 NCAS ......................................................................................................................................................................................................................................................................6
4.3 ........................................................................................................................................................................................................................................................................................................6
5 ............................................................................................................................................................................................................................................................................................... 7
5.1 74.1 PMCF ............................................................................................................................................................................................................7
5.2 .............................................................................................................................................................................................................................................................7
5.3 ..........................................................................................................................................................................................................................................7
5.4 PMCF ......................................................................................................................................................................................................................................8
6 ........................................................................................................................................................................................................................................................................................ 8
7 .................................................................................................................................................................................................................................................................................................... 8
8 ............................................................................................................................................................................................................................................................................................... 8
8.1 NCAS .............................................................................................................................................................................................................................................................................................8
8.2 ................................................................................................................................................................................................................................................................................................9
9 ............................................................................................................................................................................................................................................................................................ 9
10 .................................................................................................................................................................................................................................................................................................. 11
10.1 ................................................................................................................................................................................................................................................................................11
10.2 ........................................................................................................................................................................................................................................................................13
11 .................................................................................................................................................................................................................................................................................................. 16
12 ...................................................................................................................................................................................................................................................... 16
Page 3 of 19
摘要:
展开>>
收起<<
MDCG2020101OrH10kvlEU20177450NSuVhhN4xzvNvQhbTJ2022t10ggeNu1kv2017745Sl103gazvSuVhhSOMDCGS0MDCGu1bgbTXVvNhbu1kvYTXOvNTNhbNN0eNvkm2YTXOeNNNSfkm2YTXOvezW0geNNhvNOUpWGNQwglgRSgkm2TvlbYkvlOQQwggRv0Page1of19evxNR5194onon522102145hhQS10219201021517eNIMDCG2020101OrH1vSfQsNbTJLvfQsNXkQbTJhvffePage2of19May20...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
环境体系管理评审文件VIP免费

 2024-04-19 96
2024-04-19 96 -
管理评审控制程序(体系文件)VIP免费

 2024-04-19 106
2024-04-19 106 -
QP-05 管理评审控制程序VIP免费

 2024-04-19 150
2024-04-19 150 -
ISO17025:2017一整套程序文件(实验室认可服务)VIP免费

 2024-06-05 120
2024-06-05 120 -
医疗器械工作程序文件VIP免费
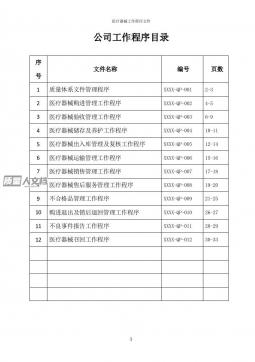
 2024-12-10 168
2024-12-10 168 -
确认与验证管理规程VIP免费

 2025-04-21 58
2025-04-21 58 -
有害物质管理程序(含表格)VIP免费

 2025-05-04 76
2025-05-04 76 -
医疗器械上市后临床跟踪控制程序VIP免费

 2025-10-27 51
2025-10-27 51 -
供应商质量管理程序VIP免费

 2025-11-07 105
2025-11-07 105 -
DC2-09001 环境因素识别、评价与控制程序VIP免费
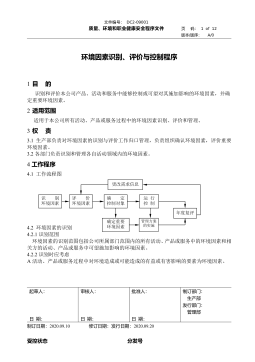
 2026-03-21 18
2026-03-21 18
作者:一如既往
分类:法规规范
价格:100质量币
属性:19 页
大小:3.03MB
格式:PDF
时间:2026-03-17


