VALCHLORgel0.016%(氮芥外用软有)药品审评(FDA)
VIP免费
CENTER FOR DRUG EVALUATIONAND
RESEARCH
APPLICATION NUMBER:
202317Orig1s000
MEDICAL REVIEW(S)
CLINICAL REVIEW
Application Type
Application Number(s)
Priority or Standard
Submit Date(s)
Received Date(s)
PDUFA Goal Date
Division/Office
Reviewer Name(s)
Review Completion Date
Established Name
(Proposed)Trade Name
Therapeutic Class
Applicant
Formulation(s)
Dosing Regimen
Indication(s)
Intended Population(s)
NDA 505(b)(2)
202317
Class 2 Resubmission
27 February 2013
27 February 2013
27 August 2013
DHP/OHOP
R.Angelo de Claro,M.D.
25 June 2013
Mechlorethamine
Valchlor
Antineoplastic
Ceptaris Therapeutics,Inc.
0.016%gel
Topical,once daily
CTCL Stage IA and IB,
second-line therapy
Adults ≥18 years
Template Version:March 6,2009
Reference ID:3331209
Clinical Review
R. Angelo de Claro , M.D.
NDA 202317
Valchlor ( Mechlorethamine gel)
Table of Contents
1 RECOMMENDATIONS /RISK BENEFIT ASSESSMENT ............................................... 5
1.1 Recommendation on Regulatory Action ......................................................................... 5
1.2 Risk Benefit Assessment ................................................................................................... 5
1.3 Recommendations for Postmarket Risk Evaluation and Mitigation Strategies ... 7
1.4 Recommendations for Postmarket Requirements and Commitment ..................... 7
2 INTRODUCTION AND REGULATORY BACKGROUND ............................................... 7
3 ETHICS AND GOOD CLINICAL PRACTICES ...................................................................
8
3.1 Submission Quality and Integrity ..................................................................................... 8
3.2 Compliance with Good Clinical Practices....................................................................... 8
3.3 Financial Disclosures........................................................................................................... 8
4 SIGNIFICANT EFFICACY /SAFETY ISSUES RELATED TO OTHER REVIEW
DISCIPLINES ...............................................................................................................................
8
4.1 Chemistry Manufacturing and Controls.............................................................................
8
4.2 Clinical Microbiology ...........................................................................................................
9
4.3 Preclinical Pharmacology /Toxicology............................................................................... 9
4.4 Clinical Pharmacology...........................................................................................................9
5 SOURCES OF CLINICAL DATA ........................................................................................... 9
5.1 Tables of Studies /Clinical Trials ....................................................................................... 9
5.2 Review Strategy ................................................................................................................... 9
5.3 Discussion of Individual Studies /Clinical Trials.............................................................. 9
6 REVIEW OF EFFICACY...............................................................................................................
9
Efficacy Summary ...........................................................................................................................
9
7 REVIEW OF SAFETY ............................................................................................................. 10
Safety Summary ........................................................................................................................... 10
7.1 Methods.................................................................................................................................. 11
7.2 Adequacy of Safety Assessments................................................................................... 11
7.3 Major Safety Results ..........................................................................................................11
8 POSTMARKET EXPERIENCE ............................................................................................... 11
9 APPENDICES ........................................................................................................................... 12
9.1 Literature Review /References ....................................................................................... 12
9.2 Labeling Recommendation .............................................................................................. 12
9.3 Advisory Committee Meeting........................................................................................... 13
2
Reference ID:3331209
摘要:
展开>>
收起<<
CENTERFORDRUGEVALUATIONANDRESEARCHAPPLICATIONNUMBER:202317Orig1s000MEDICALREVIEW(S)CLINICALREVIEWApplicationTypeApplicationNumber(s)PriorityorStandardSubmitDate(s)ReceivedDate(s)PDUFAGoalDateDivision/OfficeReviewerName(s)ReviewCompletionDateEstablishedName(Proposed)TradeNameTherapeuticClassApplicant...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
纯化水中国药典2020版与2025版GAP分析VIP免费
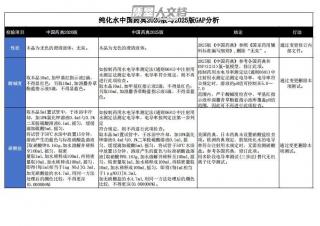
 2025-04-18 375
2025-04-18 375 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 579
2025-09-27 579 -
1101无菌检查法对比表(2025版药典 VS 2020版药典)
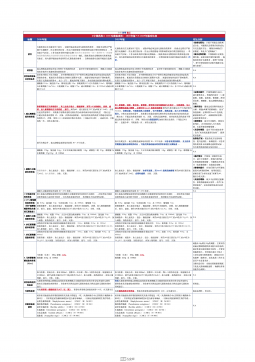
 2025-09-29 488
2025-09-29 488 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 968
2025-09-29 968 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 95
2025-11-04 95 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 506
2025-11-06 506 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 168
2025-11-06 168 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 162
2025-11-19 162 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 413
2025-11-26 413 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 76
2026-03-19 76
作者:冒牌货
分类:专业资料
价格:100质量币
属性:132 页
大小:1.21MB
格式:PDF
时间:2026-01-13


