MDR技术文档风险管理计划
VIP免费
Risk Management Plan
(According to EN ISO 14971:2019)
Product Name:
Model No.:
Document No.:
Version:
Complied by:
Reviewed by:
Authorized by:
Date:
Date:
Date:
Table of Contents
0. Revision History ......................................................................................................................................................................................... 3
1. Scope and Introduction ....................................................................................................................................................................3
1.1 Scope......................................................................................................................................................................................... 3
1.2 Introduction ............................................................................................................................................................................3
2. Definition ............................................................................................................................................................................................... 3
3. Referent Standards and Bibliography ........................................................................................................................................ 3
3.1 Referent Standards ............................................................................................................................................................. 3
3.2 Referent Document ................................................................................................................................................................ 5
4. Risk Management policy and Risk management proces ..................................................................................................6
4.1 Risk Management policy..................................................................................................................................................... 6
4.2 Risk management proces....................................................................................................................................................6
5. Risk Management plan.................................................................................................................................................................... 8
5.1 Scope of the plan ................................................................................................................................................................8
5.2 Product Information ............................................................................................................................................................... 8
5.3 Verification plan....................................................................................................................................................................10
5.4 Members and responsibilities ..................................................................................................................................... 11
5.5 Summary of review activities ......................................................................................................................................... 11
5.6 Risk assessment and risk acceptance criteria .........................................................................................................13
6 Risk Analysis and Contro ............................................................................................................................................................. 15
6.1. Identification of hazards................................................................................................................................................. 15
6.2 The initial hazard analysis , risk assessment , risk control measures and residual risk evaluation for
m 15
6.3 Conclusion of control measures ..................................................................................................................................... 17
6.4 Risk/Benefit analysis and review of overall residual risk acceptability .......................................................... 17
6.5 Adverse event report information .................................................................................................................................17
6.6 Production and post- production information .............................................................................................................17
7. Risks arising from risk control measures ..............................................................................................................................17
8. Completeness of risk contro ......................................................................................................................................................... 17
8.1 Residualriskassessment .....................................................................................................................................................17
8.2 Overall residual risk evaluation ..................................................................................................................................... 17
8.3 Residual risk information disclosure .............................................................................................................................17
9. Evaluation of overallresidualrisk acceptability ...................................................................................................................... 17
10. Production and post -production activities .........................................................................................................................17
11. Conclusion ..................................................................................................................................................................................... 17
0.Revision History
No.
Description
Approved By
State
Effective Date
1.Scope and Introduction
1.1 Scope:
1.2 Introduction:
2.Definition
o Harm:Physical injury and/or damage to health or property.
o Hazard:A potential source of harm
o Risk:The probable rate of occurrence of a hazard causing harm and the degree of severity of the
harm
o Risk Analysis:The investigation of available information to identify hazards and to estimaterisks.
o Safety:Freedom from unacceptable risk of harm
3.Referent Standards and Bibliography
3.1 Referent Standards
No
Document Number
Document Title
摘要:
展开>>
收起<<
RiskManagementPlan(AccordingtoENISO14971:2019)ProductName:ModelNo.:DocumentNo.:Version:Compliedby:Reviewedby:Authorizedby:Date:Date:Date:TableofContents0.RevisionHistory......................................................................................................................................
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
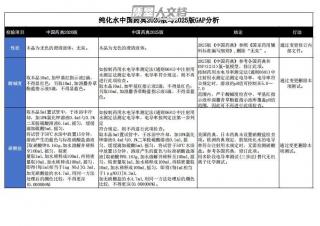
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 375
2025-04-18 375 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 579
2025-09-27 579 -
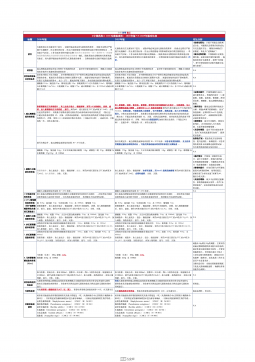
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 488
2025-09-29 488 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 968
2025-09-29 968 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 95
2025-11-04 95 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 506
2025-11-06 506 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 168
2025-11-06 168 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 162
2025-11-19 162 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 413
2025-11-26 413 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 76
2026-03-19 76
作者:冒牌货
分类:专业资料
价格:60质量币
属性:15 页
大小:354.37KB
格式:PDF
时间:2026-01-14


