FDA:磁共振(MR)环境中医疗器械安全性的测试与标签_中英文版
VIP专享
Contains Nonbinding Recommendations
Testing and Labeling Medical
Devices for Safety in the Magnetic
Resonance (MR) Environment
Guidance for Industry and Food and
Drug Administration Staff
Document issued on October 10, 2023.
Document originally issued on May 20, 2021
For questions about this document, contact the Office of Science & Engineering Laboratories
(OSEL), OSEL_CDRH@fda.hhs.gov, (301) 796-2530.
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Devices and Radiological Health
Contains Nonbinding Recommendations
Preface
Public Comment
You may submit electronic comments and suggestions at any time for Agency consideration
to https://www.regulations.gov. Submit written comments to the Dockets Management Staff,
Food and Drug Administration, 5630 Fishers Lane, Room 1061, (HFA-305), Rockville, MD
20852-1740. Identify all comments with the docket number FDA-2019-D-2837. Comments
may not be acted upon by the Agency until the document is next revised or updated.
Additional Copies
Additional copies are available from the Internet. You may also send an e-mail request to
CDRH-Guidance@fda.hhs.gov to receive a copy of the guidance. Please use the document
number GUI01500059 and the complete title of the guidance in the request.
摘要:
展开>>
收起<<
ContainsNonbindingRecommendationsTestingandLabelingMedicalDevicesforSafetyintheMagneticResonance(MR)EnvironmentGuidanceforIndustryandFoodandDrugAdministrationStaffDocumentissuedonOctober10,2023.DocumentoriginallyissuedonMay20,2021Forquestionsaboutthisdocument,contacttheOfficeofScience&EngineeringLab...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
纯化水中国药典2020版与2025版GAP分析VIP免费
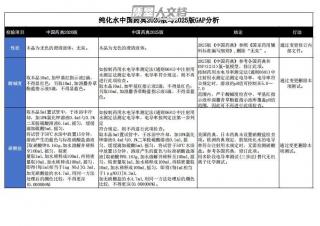
 2025-04-18 375
2025-04-18 375 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 579
2025-09-27 579 -
1101无菌检查法对比表(2025版药典 VS 2020版药典)
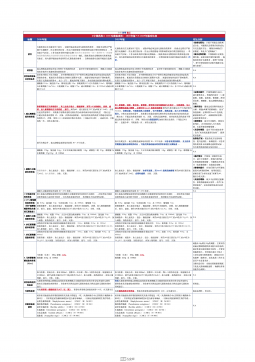
 2025-09-29 488
2025-09-29 488 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 968
2025-09-29 968 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 95
2025-11-04 95 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 506
2025-11-06 506 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 168
2025-11-06 168 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 162
2025-11-19 162 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 413
2025-11-26 413 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 76
2026-03-19 76
作者:薛定谔的龙猫
分类:专业资料
价格:100质量币
属性:64 页
大小:1.78MB
格式:PDF
时间:2025-11-11


