FDA行业指南:有关食品过敏原标签的问答(第 5 版)_中英文
VIP专享
Contains Nonbinding Recommendations
1
Questions and Answers Regarding Food
Allergens, Including the Food Allergen
Labeling Requirements of the Federal
Food, Drug, and Cosmetic Act
(Edition 5): Guidance for Industry
You can comment on any guidance at any time (see 21 CFR 10.115(g)(5)). Submit electronic
comments to https://www.regulations.gov. Submit written comments to the Dockets
Management Staff (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061,
Rockville, MD 20852. All comments should be identified with the docket number FDA-
2022-D-0099.
For questions regarding this document, contact the Human Foods Program at
HFP-Policy@fda.hhs.gov.
Additional copies are available at https://www.fda.gov/FoodGuidances.
U.S. Department of Health and Human Services
Food and Drug Administration
Human Foods Program
January 2025
OMB Control No. 0910-0792
Current expiration date available at https://www.reginfo.gov/public/do/PRAMain
See additional PRA statement in Section III of this guidance.
包含无约束力的建议
1
关于食品过敏原的问题和答案,包括
《联邦食品、药品和化妆品法案》
(第5版)的食品过敏原标签要求:行
业指南
https://
www.regulations.gov
您可随时对任何指南提出意见(参见21 CFR 10.115(g)(5)),电子意见请提交至
,书面意见请提交至文件管理工作人员(HFA -305),美国食品
药品监督管理局,5630 Fishers Lane,Rm. 1061,Rockville,MD 20852。所有意见均应
注明案卷号FDA-2022-D-0099。
HFP-Policy@fda.hhs.gov如对本文件有疑问,请联系人类食品项目 。
https://www.fda.gov/FoodGuidances
其他副本可在 处获得。
美国卫生与公众服务部
美国食品药品监督管理局
人类食品计划
2025年1月
OMB控制编号0910-0792
https://www.reginfo.gov/public/do/PRAMain当前有效期可至 ,详见本指南第
三部分的 PRA 补充说明。
Contains Nonbinding Recommendations
2
Table of Contents
I. Introduction .................................................................................... 3
II. Questions and Answers .................................................................. 4
A. General Information……………………………………………………………………. 4
B. Types of Foods That Fall Under the Food Allergen Labeling Requirements of the
Federal Food, Drug, and Cosmetic Act……………………………………………………. 6
C. Food Sources…………………………………………………………………………... 13
D. The Food Allergen Labeling Requirements of the Federal Food, Drug, and Cosmetic
Act………………………………………………………………………………………… 16
E. Dietary Supplements…………………………………………………………………... 24
III. Paperwork Reduction Act of 1995 .............................................. 26
摘要:
展开>>
收起<<
ContainsNonbindingRecommendations1QuestionsandAnswersRegardingFoodAllergens,IncludingtheFoodAllergenLabelingRequirementsoftheFederalFood,Drug,andCosmeticAct(Edition5):GuidanceforIndustryYoucancommentonanyguidanceatanytime(see21CFR10.115(g)(5)).Submitelectroniccommentstohttps://www.regulations.gov.Su...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
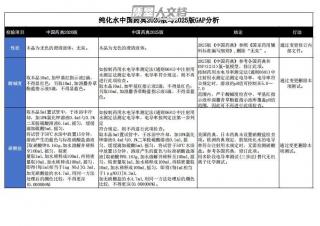
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 398
2025-04-18 398 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 655
2025-09-27 655 -
1101无菌检查法对比表(2025版药典 VS 2020版药典)
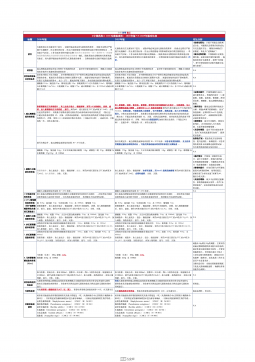
 2025-09-29 600
2025-09-29 600 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 999+
2025-09-29 999+ -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 549
2025-11-06 549 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 187
2025-11-06 187 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 207
2025-11-19 207 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 512
2025-11-26 512 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 100
2026-03-19 100 -
PDA 技术报告第60号(修订版2026):工艺验证:生命周期方法

 2026-03-19 310
2026-03-19 310
作者:薛定谔的龙猫
分类:专业资料
价格:100质量币
属性:54 页
大小:1.19MB
格式:PDF
时间:2025-11-12


