ISO 10993-13 2010医疗器械的生物学评价第 13部分基于聚合物的医疗器械降解产物的识别与定(中文)
VIP免费
ISO 1
0993-13
2010
6 1
5
13
ISO 2010
Reference number
ISO 10993-13:2010(E)
10993-13:2010(E)ISO
PDF
PDF Adobe
Adobe
Adobe Adobe Systems Incorporated
PDF PDF
ISO
ISO 2010
ISO
ISO ISO
56CH-1211 20
+4122749011
+41 22 749 09
47 copyright@
iso.org www.iso.
org
2ISO 2010-All rights reserved
ISO 10993-13:2010(E)
4
........................................................................................................................................................................................................................
1 ......................................................................................................................................................................................................................1
2 1
3 ............................................................................................................................................................................. 2
4 ..................................................................................................................................................................... 2
4.1 2
4.2 ..................................................................................................................................................................5
4.3 ..................................................................................................6
5 .............................................................................................................................................................................................6
5.1 ............................................................................................................................................................................................................... 6
5.2 ....................................................................................................................................................... 6
5.3 6
5.4 .................................................................................................9
6 ............................................................................................................................................................................................................10
.............................................................................................................................................................1
B Tg12
........................................................................................................................................................................................................................14
iii2010- All rights reserved
ISO
摘要:
展开>>
收起<<
ISO1099313VEhQNrH2010t6g15eSuVhhvurifNSANRZTriSuVhhMNrivtNRgSuVhhvurifN13RWNZTrivSuVhhMNrivRNISO2010ReferencenumberISO10993132010E10993132010EISOPDFQMXfkdPDFeNSSTLQeWOS0h9cnAdobevSeVkdeNSbSSpbgwOFdLQevWOSSvWbgLdOvgNTRNL0NkdeNvTeWkdbbNSAdobeSeVvN0VEhQSNYyNfYkdNbbNOUN0AdobefAdobeSystemsIncorporatedvUF...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
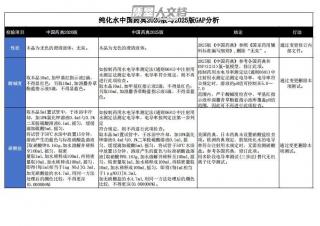
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 370
2025-04-18 370 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 578
2025-09-27 578 -
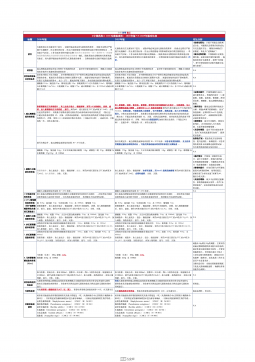
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 486
2025-09-29 486 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 966
2025-09-29 966 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 94
2025-11-04 94 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 506
2025-11-06 506 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 168
2025-11-06 168 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 162
2025-11-19 162 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 409
2025-11-26 409 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 73
2026-03-19 73
作者: 51zlzl
分类:法规规范
价格:200质量币
属性:20 页
大小:2.54MB
格式:PDF
时间:2025-11-20


