PDA TR22-2025-Process-Simulation-for-Aseptically-Filled-Products
VIP免费
pda.org
Technical Report
No.
Process Simulation for
Aseptically Filled
©PDA 2025
Products
22
Revised 2025
Technical Report No. 22 (Revised 2025) ©2025Parenteral DrugAssociation, Inc.
Technical Report
No.
i
22
Process Simulation for
Aseptically Filled
Products
Revised 2025
ISBN: 978-1-945584-55-8
© 2025Parenteral Drug Association, Inc.
All rights reserved
Technical Report No. 22 (Revised 2025) © 2025 Parenteral Drug Association, Inc.
ii
Authors
Co-Leads
Marcia Baroni
Emergent BioSolutions, Inc.
Gabriele Gori
Chiesi Pharmaceuticals SpA
Harold S. Baseman
ValSource, Inc.
Subrata Chakraborty, PhD
Inophar Consulting and Training
Biswarup DasGupta
Vertex Pharmaceuticals
Michael Dekner
Independent Expert
Caroline Elsabe Dreyer
Novo Nordisk
Vanessa Vasadi Figueroa
VVF Science, LLC
Benoit Franquin
CSL Behring
Guenther Gapp
Gapp Quality GmbH
Ian Hudson
Thermo Fisher Scientific
Stephen E. Langille, PhD
ValSource, Inc.
Morcos Loka
Minapharm
James Matthews
Cardinal Health
William Miele
Pfizer
Patrizia Muscas, PhD
Eli Lilly
Darius Pillsbury
ValSource, Inc.
Christine Sherman
Takeda Manufacturing USA, Inc.
摘要:
展开>>
收起<<
pda.orgTechnicalReportNo.ProcessSimulationforAsepticallyFilled©PDA2025Products22Revised2025TechnicalReportNo.22(Revised2025)©2025ParenteralDrugAssociation,Inc.TechnicalReportNo.i22ProcessSimulationforAsepticallyFilledProductsRevised2025ISBN:978-1-945584-55-8©2025ParenteralDrugAssociation,Inc.Allrigh...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
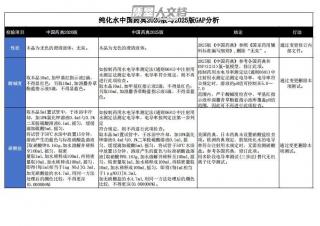
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 383
2025-04-18 383 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 608
2025-09-27 608 -
1101无菌检查法对比表(2025版药典 VS 2020版药典)
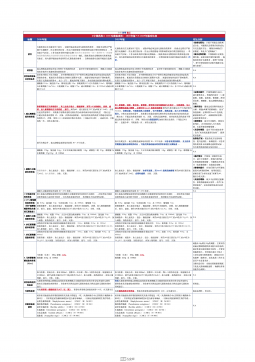
 2025-09-29 557
2025-09-29 557 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 999+
2025-09-29 999+ -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 520
2025-11-06 520 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 176
2025-11-06 176 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 186
2025-11-19 186 -
注射用药品的目视检查-第五版-2025.10_中文VIP免费

 2025-11-25 82
2025-11-25 82 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 438
2025-11-26 438 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 85
2026-03-19 85
作者: 51zlzl
分类:专业资料
价格:50质量币
属性:130 页
大小:4.46MB
格式:PDF
时间:2025-11-18


