ISO 10555-6 2015 Amd 1 2019
VIP免费
INTERNATIONAL
STANDARD
ISO
10555-6
First edition
2015-04-15
AMENDMENT 1
2019-09
Intravascular catheters—Sterile and
single-use catheters—
Part 6:
Subcutaneous implanted ports
AMENDMENT 1
Cathéters intravasculaires—Cathéters stériles et non réutilisables—
Partie 6:Chambres à cathéter implantables
AMENDEMENT 1
Reference number
ISO 10555-6:2015/Amd.1:2019(E)
C ISO 2019
ISO10555-6:2015 /Amd.1:2019(E)
COPYRIGHT PROTECTED DOCUMENT
◎ISO 2019
All rights reserved.Unless otherwise specified,or required in the context of its implementation,no part of this publication may
be reproduced or utilized otherwise in any form or by any means,electronic or mechanical,including photocopying,or posting
on the internet or an intranet,without prior written permission.Permission can be requested from either ISO at the address
below or ISO's member body in the country of the requester.
ISO copyright office
CP 401·Ch.de Blandonnet 8
CH-1214 Vernier,Geneva
Phone:+41227490111
Fax:+41227490947
Email:copyright@iso.org
Website:www.iso.org
Published in Switzerland
ii ◎ISO 2019-Allrights reserved
ISO 10555-6:2015/Amd.1:2019(E)
Foreword
ISO(the International Organization for Standardization)is a worldwide federation of national standards
bodies (ISO member bodies).The work of preparing International Standards is normally carried out
through ISO technical committees.Each member body interested in a subject for which a technical
committee has been established has the right to be represented on that committee.International
organizations,governmental and non-governmental,in liaison with ISO,also take part in the work.
ISO collaborates closely with the International Electrotechnical Commission (IEC)on all matters of
electrotechnical standardization.
The procedures used to develop this document and those intended for its further maintenance are
described in the ISO/IEC Directives,Part 1.In particular,the different approval criteria needed for the
different types of ISO documents should be noted.This document was drafted in accordance with the
editorial rules of the ISO/IEC Directives,Part 2(see www.iso.org/directives ).
Attention is drawn to the possibility that some of the elements of this document may be the subject of
patent rights.ISO shall not be held responsible for identifying any or all such patent rights.Details of
any patent rights identified during the development of the document will be in the Introduction and/or
on the ISO list of patent declarations received (see www.iso.org/patents).
Any trade name used in this document is information given for the convenience of users and does not
constitute an endorsement.
For an explanation of the voluntary nature of standards,the meaning of ISO specific terms and
expressions related to conformity assessment,as well as information about ISO's adherence to the
World Trade Organization (WTO) principles in the Technical Barriers to Trade (TBT),see www.iso
.org/iso/foreword.html .
This document was prepared by Technical Committee ISO/TC84,Devices for administration of medicinal
products and catheters.
A list ofall parts in the ISO10555 series can be found on the ISO website.
Any feedback orquestions on this document should be directed to the user's national standards body.A
complete listing of these bodies can be found atwww.iso.org/members.html .
◎ISO 2019-All rights reserved iii
标签: #ISO
摘要:
展开>>
收起<<
INTERNATIONALSTANDARDISO10555-6Firstedition2015-04-15AMENDMENT12019-09Intravascularcatheters—Sterileandsingle-usecatheters—Part6:SubcutaneousimplantedportsAMENDMENT1Cathétersintravasculaires—Cathétersstérilesetnonréutilisables—Partie6:ChambresàcathéterimplantablesAMENDEMENT1ReferencenumberISO10555-6...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
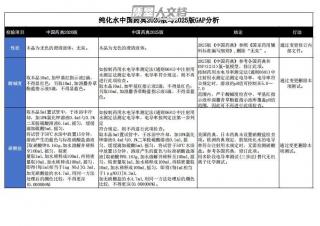
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 381
2025-04-18 381 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 585
2025-09-27 585 -
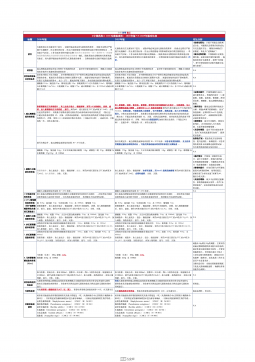
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 516
2025-09-29 516 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 989
2025-09-29 989 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 102
2025-11-04 102 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 510
2025-11-06 510 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 171
2025-11-06 171 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 164
2025-11-19 164 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 424
2025-11-26 424 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 80
2026-03-19 80
作者:安心365
分类:法规规范
价格:10质量币
属性:6 页
大小:197.38KB
格式:PDF
时间:2026-05-08


