202511PDA CCS的根本原因分析_(中英文)
VIP专享
pda.org
©PDA 2025
pda.org
©PDA 2025
PDA From Findings to Fixes: Strengthening CCS in Aseptic Manufacturing
Workshop 2025
Root Cause Analysis of
a CCS
David Keen
个人数字助理
©PDA 2025
PDA从发现到修复:加强无菌生产中的中国化学会
2025年研讨会
大卫·基
CCS的根本原因分析
恩
pda.org
©PDA 2025
Introduction
When is a contamination failure a CCS failure?
CCS is a continuous feedback loop that tells you about the health of your manufacturing system
Your CCS should have metrics for control/not in control for each Critical Control Point (CCP)
You should know what your CCPs are
What happens when something goes wrong and your CCS says everything is OK?
Have you missed a control point in the CCS?
Has the CCS failed?
摘要:
展开>>
收起<<
pda.org©PDA2025pda.org©PDA2025PDAFromFindingstoFixes:StrengtheningCCSinAsepticManufacturingWorkshop2025RootCauseAnalysisofaCCSDavidKeen个人数字助理©PDA2025PDA从发现到修复:加强无菌生产中的中国化学会2025年研讨会大卫·基CCS的根本原因分析恩pda.org©PDA2025IntroductionWhenisacontaminationfailureaCCSfailure?CCSisacontinuousfeedbackloopthattellsyo...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
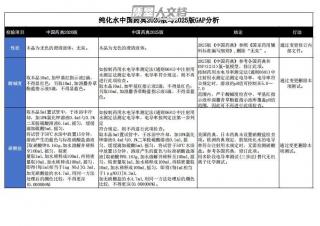
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 375
2025-04-18 375 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 579
2025-09-27 579 -
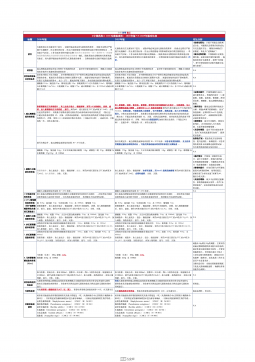
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 488
2025-09-29 488 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 968
2025-09-29 968 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 95
2025-11-04 95 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 506
2025-11-06 506 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 168
2025-11-06 168 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 162
2025-11-19 162 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 413
2025-11-26 413 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 76
2026-03-19 76
作者: 51zlzl
分类:专业资料
价格:60质量币
属性:84 页
大小:539.75KB
格式:PDF
时间:2025-11-21


