PDA 先进治疗药物产品生产-设施设计(第一部分)Points to Consider No. 14 Manufacturing of ATMPs - Facility Design (Part 1) 中英文
VIP免费
1
PTC NO.14 先进治疗药物产品生产
——设施设计(第一部分)
Points to Consider No.14
Manufacturing of ATMPs -Facility Design
(Part 1)
先进治疗药物产品生产
——设施设计(第一部分)
2
Authors
Lead
Richard Denk
SKAN AG
Rebecca Brewer
TM Pharma Group
Quality Executive Partners
Subrata Chakraborty
Gxpfont
Palash Das
Baxter
Alberto Gonzales
Takeda
Mark Hallworth
Particle Measuring
Rakesh Jakkashetty
SaudiBio
Lizette Janse van Rensburg
Eurolab
Claudia Lee
Velvet Therapeutics
Gerry McKiernan
CellTherapie
Juergen Metzger
Sartorius
Tracy Moore
Youwen Pan
WuXi Biologics
Jean-Sebastien Parisse
Aseptic Technologies
David Phasey
3P innovation
Durga Prasad Madhavapeddi
FUJIFILM Diosynth Biotechnologies
John Rapi
NNIT
Gilli Rosenzweig
Avrobio
Osamu Shirokizawa
Life Scientia,Ltd.
Andiyanto Sutandar
NNIT
Tricia Vail
Sartorius
Hartmut F.Zimmerman
Boehringer Ingelheim
Manufacturing of ATMPs-
Facility Design (Part 1)
Points to Consider No.14
2025
ISBN:978-1-945584-51-0
◎
2025 Parenteral Drug Association,Inc.All rights reserved.
KRBConsu
KRBCo
3
摘要:
展开>>
收起<<
1PTCNO.14先进治疗药物产品生产——设施设计(第一部分)PointstoConsiderNo.14ManufacturingofATMPs-FacilityDesign(Part1)先进治疗药物产品生产——设施设计(第一部分)2AuthorsLeadRichardDenkSKANAGRebeccaBrewerTMPharmaGroupQualityExecutivePartnersSubrataChakrabortyGxpfontPalashDasBaxterAlbertoGonzalesTakedaMarkHallworthParticleMeasuringRakeshJakkashe...
声明:如果您的权利被侵害,请联系我们的进行举报。
相关推荐
-
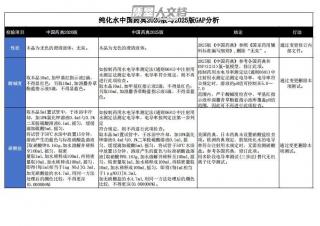
纯化水中国药典2020版与2025版GAP分析VIP免费
 2025-04-18 375
2025-04-18 375 -
无菌检查法对比表(2025版药典 VS 2020版药典)

 2025-09-27 579
2025-09-27 579 -
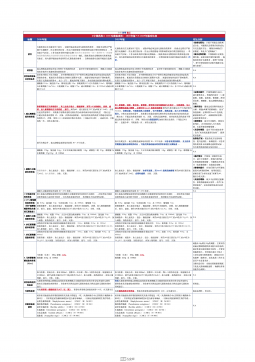
1101无菌检查法对比表(2025版药典 VS 2020版药典)
 2025-09-29 488
2025-09-29 488 -
1143 细菌内毒素检查法对比表(2025版药典 VS 2020版药典)

 2025-09-29 968
2025-09-29 968 -
ECA-污染控制策略指南(中英文)-202202VIP免费

 2025-11-04 95
2025-11-04 95 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(中英文对照版)VIP专享

 2025-11-06 506
2025-11-06 506 -
TR-26-2025-Sterilizing-Filtration-of-Liquids液体的灭菌过滤(英文版)VIP免费

 2025-11-06 168
2025-11-06 168 -
PDA-TR49-生物清洁验证-中英文翻译

 2025-11-19 162
2025-11-19 162 -
硫酸艾玛昔替尼片(CXHS2300097)说明书VIP免费

 2025-11-26 413
2025-11-26 413 -
PDA TR60 Process Validation A Lifecycle Approach 2026VIP免费

 2026-03-19 76
2026-03-19 76
作者: 51zlzl
分类:专业资料
价格:1质量币
属性:78 页
大小:1.37MB
格式:PDF
时间:2025-11-25


